Regenerative therapies are non-surgical, minimally invasive and offer faster relief from pain and discomfort. A range of treatments each complementing the other to accelerate healing can be administered after assessment of injury and health history. At StemX, we provide the following treatments:
Stem Cells and Growth Factors from Wharton's Jelly
Used to augment healing of musculoskeletal injuries.

What is it
A gelatinous combination of rich substances like growth factors, exosomes, stem cells, derived from the umbilical cord after a healthy delivery. These elements play a crucial role in intercellular communication and transmission of macromolecules between cells, thereby facilitating repair of damaged tissue in the body and rebuilding compromised cell structures.
How is it administered?
This treatment option normally consists of a singular injection. Results are visible in 6 months but the patient might notice progress in 2-3 months. This treatment is not age-dependent.
What is it derived from?
Umbilical cord cells of healthy, consenting mothers.Hyaluronic Acid Injections
Used to help increase lubrication and decrease pain in osteoarthritic knees.
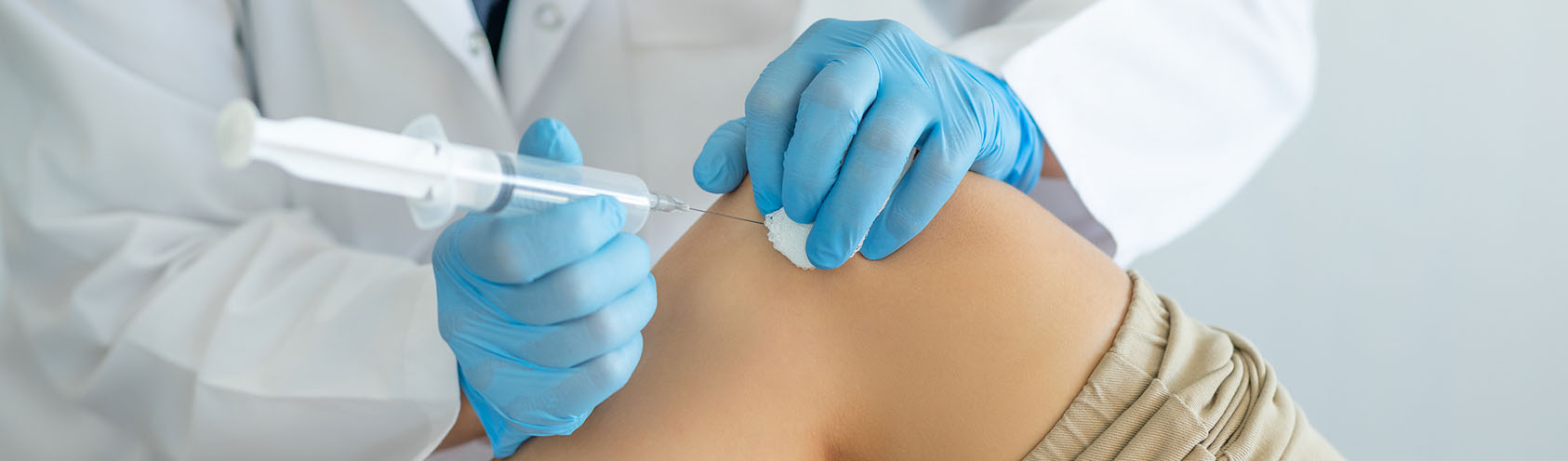

What is it
Hyaluronic Acid (HA) is a naturally occurring molecule in the body, mostly found in the skin, within eye sockets and other joints and tissue where its chief function is to increase moisture and provide flexibility to the surrounding areas. It is typically injected into the affected area.
How is it administered?
HA injections are injected into joints. This treatment initially consists of a 3 part series one week apart. This is mostly used in combination with either Wharton's Jelly, and Growth Factor.
What is it derived from?
Manufactured in an FDA approved lab.Platelet Rich Plasma (PRP) Therapy
Used to trigger cell production and stimulate healing of injured tendons, ligaments, muscles and joints.


What is it
Platelet rich plasma (PRP) consists of plasma and platelets, which contain growth factors with can be injected in affected areas to increase the concentration of specific bioproteins or hormones. Chronic joint injuries can take a long time to heal so adding PRP injections as a part of the treatment can augment the healing process and restore mobility and function.
How is it administered?
After creating platelet rich plasma from the patient's blood sample, the solution is injected into the affected area using ultrasound imaging for guidance. This treatment is typically offered to patients under the age of 50. It takes 2-3 treatments for pain relief.
What is it derived from?
Injections are prepared from a sample of the patients own blood.Factors To Consider With Regenerative Medicine Treatments
Some prescription drugs and lifestyle behaviors might interfere with stem cell production and healing through advanced regenerative medicine. We recommend avoiding the following:
- Non-Steroidal Anti-Inflammatory Drugs (NSAIDS) such as ibuprofen, Aleve, Motrin, Mobic, Celebrex, etc
- Aspirin and steroids
- Alcohol
- Tobacco
Patients with severe anemia, low platelet count, abnormal platelet function, active systemic infection, or active cancer are not ideal candidates for regenerative medicine.
We will discuss any concerns prior to treatment.
Do you need help with
Chronic joint pain?
Are you interested in a one time, non-invasive procedure to improve function and mobility? Non-surgical, non-invasive therapy for joints and cartilage is available! Find out if you are a candidate today.




